Electric Boat Basics, Part 9: Battery Chemistry

Report by Jeff Butler, Courtesy of Plugboats.com
When discussing battery chemistry, we are referring to the materials involved in the chemical reaction that produces the electric charge. There are two base materials used for the rechargeable batteries you will want for an electric boat: Lead and Lithium. Within each type of battery, there are many options.
The chemical reaction in lead batteries takes place between electrodes made of lead (the anode is lead metal and the cathode is lead oxide) and an acid. Hence the name lead-acid battery.
Lithium batteries use a variety of materials and alloys to cause the chemical reaction. The specific combination is usually available in the user manuals and brochures, but is often not advertised. The generic Lithium-ion or Li-ion is used.
The one type of lithium battery that usually has the specific chemistry in the name is the LiFePO or LiFePO4 battery, which is not lithium ion but lithium iron phosphate.

For lithium batteries there is a six-point system that gives ratings on:
- Specific Energy
- Specific Power
- Safety
- Performance
- Life Span
- Cost
We have used the system to provide a basis of comparison for the most common lithium chemistries and have adapted it slightly for lead-acid batteries to give an indication of where they fit in the scheme of things.
The 6 Rating Criteria
Specific Energy can also be called gravimetric energy density and refers to the amount of energy that can be stored in the battery by weight: watt hours per kilogram.
The term energy density is often used, but isn’t entirely accurate because energy density actually refers to how much energy can be stored by volume: kWh per liter. This by-volume spec is rarely used in consumer information, so if you see energy density it probably (but not always) means the by weight specific energy specification.

A high specific energy or energy density is better: It translates to a lighter battery. For lithium batteries this number could be anywhere from 100 watt hours per kilogram to 250 watt hours per kilogram, depending on the chemistry. Specific energy ratings for lithium batteries are 100 to 250 watt hours, not kiloWatt hours.
The lithium rating in kWh would be .1 to .25 kWh per kilogram of battery material. Lead acid batteries have specific energy of about 35-40 watt hours per kilogram (.03 kWh/kg).
The numbers for lithium refer to the energy of an individual cell. The energy density of the battery pack as a whole will be lower because it needs to take into account the weight of all the connections and coolants and glues that go into constructing the pack. The lead acid numbers are for the battery pack.

Specific Power is the amount of power per weight that the chemistry can deliver: kW per kilogram.
This is easy to understand now that we know the difference between energy and power. Lithium batteries can range from about 250 watts per kilogram to 350 w/kg. Lead acid is about 180w/kg.
Safety. This specification on the chart is the safety compared to other lithium batteries.
When it comes to concerns about battery safety, some people point to news reports of electric-vehicle fires. The reason there are news reports about EV battery fires is the same reason there are news reports about airplane accidents. They are rare. Nobody reports on the millions of hours of battery use that go on every day with no incident.

There is certainly some risk involved in using lithium-ion batteries, just as there is some risk involved in using the volatile, explosive liquids that run fossil fuel motors. When batteries are used and maintained properly the risks are low. They are mitigated by thousands of hours of research and testing as well as the battery management system monitoring that goes on every second the battery is in use.
Performance in the graphs relates to performance at hot and cold temperatures.
Life span is an indication of the overall life span of the battery and specifically how many charging cycles it can go through.
Every discharge and recharge — called a cycle — results in the battery not being able to get all the way back to fully charged. Eventually the battery is not useful. More cycles is better because it means your battery will last longer.
Determining how many cycles a battery can undergo is difficult because the definition of a cycle isn’t absolute in terms of how much energy is discharged and recharged to make up ‘a cycle.’
In general, though, lithium-ion batteries in electric vehicles and boats are designed to undergo 1,000 to 2,000 charge cycles.

Cost refers to relative initial cost compared to other lithium batteries. A note on looking at the graphs: the ratings show the desirability of the battery characteristics, so a low cost is higher on the rating scale because it is a good thing in a battery.
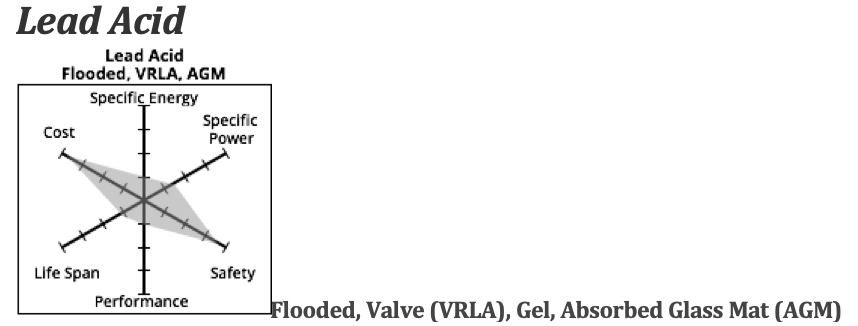
Lead Acid
A starter battery is only used to start a motor, which requires more power, but for a short period of time: small voltage and high maximum amps. It doesn’t need big energy storage. It uses a small discharge to turn the massive weight of the fossil fuel motor, then the alternator (which is just a small electricity generating turbine) recharges the battery through the spinning of the fossil fuel motor.
For an electric boat, we want a battery that has a power output (Volts times Amps) compatible with the motor and lots of available kiloWatt hours to fully discharge the battery (up to about 80%) pulling electricity out of it for a long time.
This is called a deep cycle or sometimes a deep-discharge lead-acid battery.
There are a few types of lead acid deep cycle batteries — flooded batteries and valve regulated lead acid (VRLA). The VRLAs are further divided into gel batteries and Absorbed Glass Mat (AGM) types.
In the flooded versions the electrolyte is liquid, in gel versions it is a gel and in AGM it is held in a semi solid state by being held in the pores of the glass mat.
Flooded are the heaviest and least expensive…AGM the lightest and most expensive, but still much cheaper than lithium batteries.

Compared to lithium batteries, Lead Acid batteries have lower specific energy, lower cycles and lower initial cost. The specific energy is about half of the highest-density lithium batteries — and the specific power is about half that of lithium batteries. They generally are safe, however the flooded versions can release hydrogen gas and explode under certain conditions, especially if the owner has been a bit lax in maintenance.
The initial cost for Lead Acid is about 1/4 the price of a lithium battery, but since they last for only 300-400 cycles, the cost per cycle is higher. They are good for smaller motors, but after about 48 volts, the amount of power needed to move the weight of the battery pack alone makes it an impractical choice.
Lithium
There are many lithium-battery chemistries, but some are used for things like laptops and medical devices.

Below are the main varieties used for electric boat propulsion. The chemical composition refers to the materials used in the cathodes.

NMC is the most popular chemistry for electric powertrains in EVs, e-bikes and electric boats. The main reason for its popularity is that it has a high specific energy. Different manufacturers have their own recipes for the nickel, manganese and cobalt proportions so some will sacrifice a little specific energy for more specific power and vice versa. Battery manufacturers are trying to use less cobalt because of cost and concerns about ethical mining. More nickel means higher energy density, lower cost and longer cycle life, but a slightly lower voltage.
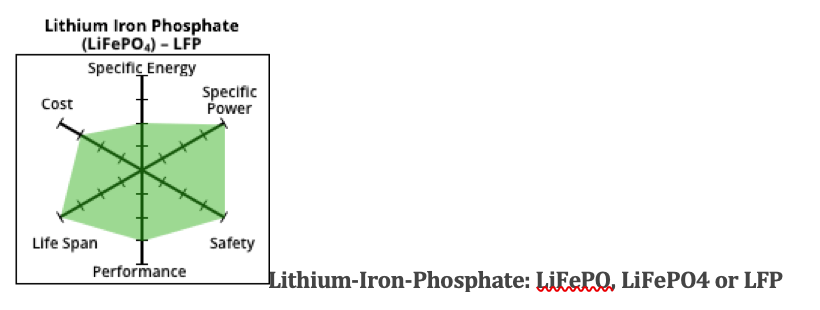
Many electric-boat owners and companies prefer the LiFePO lithium iron battery chemistry to lithium ion, largely because of lower cost and concerns about the safety of li-ion.

They make a tradeoff in range, though, because LiFePO has a low specific energy. It may not be the best battery choice for a high-speed, high-power electric boat. It is, however, becoming increasingly popular with sailboat owners where the increased battery weight in relation to the size of the boat is less important and speed under power is a non-issue.
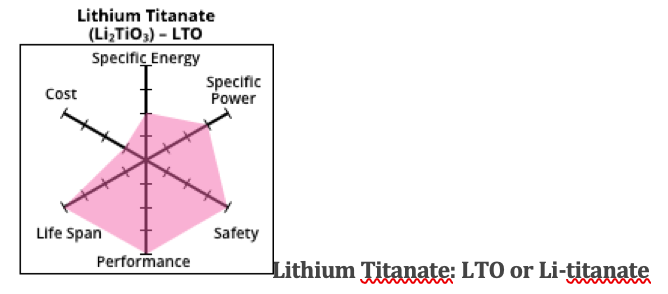
Lithium Titanate is a good chemistry for certain large vessels, but is not a popular option for recreational boats. It has a low specific energy — so batteries are heavy. On the other hand, they have long life span, are safe and can be charged quickly. The other drawback is their expense.

For a big electric ferry where the weight of the battery is less important but quick charging and public safety are important, the higher initial cost is offset by the long life span. This means the operator is free from the ongoing high operating costs of constantly filling up with fossil fuel.
Article courtesy of plugboats.com.