A Tutorial on Electrical Propulsion for Boats

A Tutorial on Electrical Propulsion for Boats.
It’s helpful to have a quick review of physics because it helps in understanding the electricity aspect of propulsion.
In the end, the distance and speed you will be able to travel in your electric boat and the relationship of the two all come down to the constraints of physics: how much energy and power (which are two different things, as you will find out) it takes to move the weight of your boat against the resistance and force of the water.
Every day we all throw around phrases like ‘I have a lot of energy’ … ‘does the government have the power to do that’ … but when it comes to figuring out what you need for your boat, physics has very specific definitions for those terms.

ENERGY: Energy is the ability of something to do work.
FORCE: Is a push or pull upon an object resulting from an interaction with another object.
WORK: Work is done when energy is transferred to an object that causes movement of the object.
POWER: Power is the rate at which the work is done.
The equations are:
Work = Force X Distance
Power = Work ÷ Time.
- Work is measured in Joules.
- Energy is measured in Joules.
- Force is measured in Newtons and is related to the weight of the thing that is being pushed or pulled.
- Distance is measured in meters.
- Power is measured in Watts.
- Time is measured in seconds.
The reason energy and work are both measured in Joules is that the work is defined as the thing that is accomplished through the transfer of the energy, so they are measured the same way.
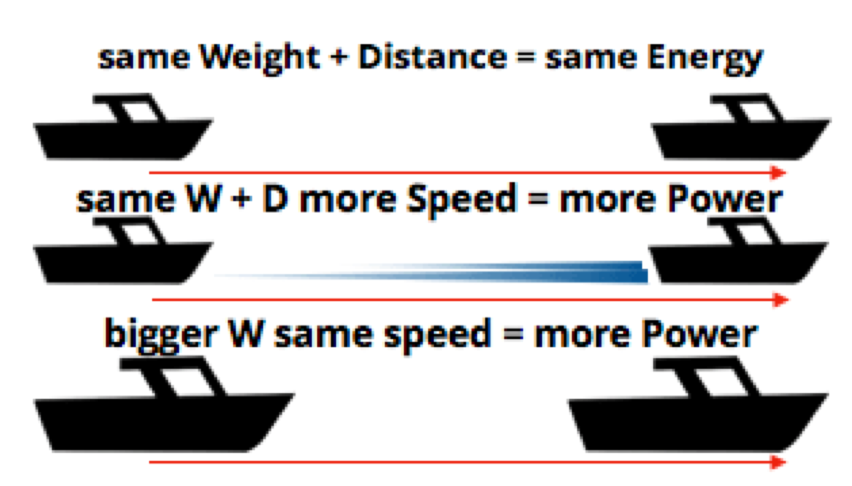
Measuring the Energy Required for Given Work
The important concept here is that moving the set weight of your boat at a set distance will always require the same amount of energy – the same amount of work (in calm water). If you want to move it the same distance faster, or move a bigger boat the same distance it will require more power:
Moving a boat through waves is essentially the same as moving a heavier boat, you’re overcoming the force of the waves, so you require more force and power to move at speed. You already know this intuitively, but it is good to know how it is all measured, as you’ll see when it comes to batteries.
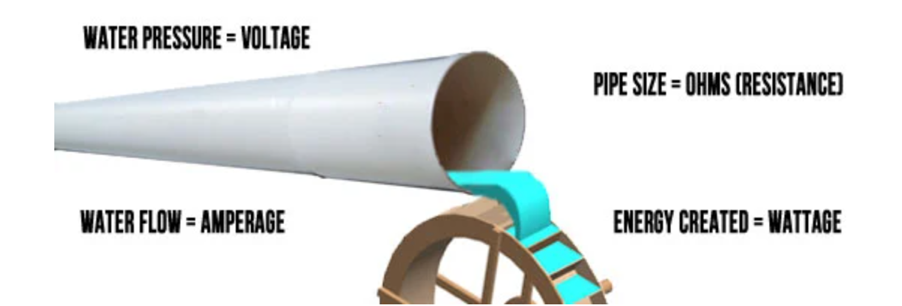
Electricity: Volts, Amps, Watts
Volts, Amperes and Watts are examples above are all explanations of mechanical energy, things moving in space. Electrical energy has its own measurements, which are intertwined with these units of mechanical energy. The three basic electrical energy terms you need to know are Volts, Amperes (amps) and Watts.
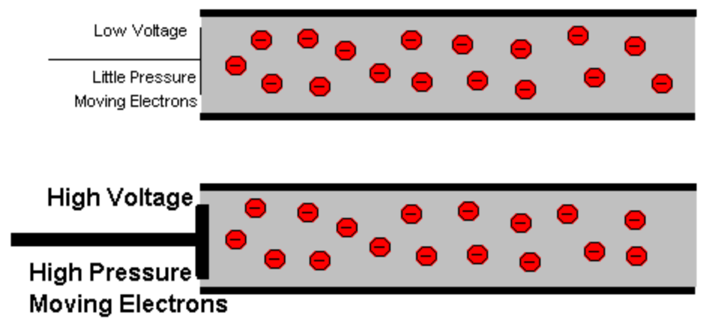
Volts (denoted as V) are the units used to measure electrical energy. To be precise, Volts measure electric potential. Electric potential is how much electric energy is capable of moving from one point to another – that idea of ‘the ability to do work’ applied to electricity. It’s not an exact analogy, but if you think of electricity as water, voltage is the water pressure.
Amps (denoted as A) are the units used to measure the flow of the energy. If voltage is the water pressure, imagine a meter on the water pipe that measures how much water, how many water molecules, are passing by a point on the pipe at any one time. With electricity, the meter is measuring the individual electric charges that are going through a wire, and the flow, the current, is measured in Amps.
Watts (denoted as W) is used to measure the electric power. Watts are the units used to measure both electrical power and mechanical power. A Watt is the unit of electrical power equal to one ampere under the pressure of one volt. One Watt is a small amount of power.
A 60-Watt light bulb uses 60 Watts per hour or 60 Joules of energy. That is .06 kiloWatts per hour.
This comes in handy when trying to figure out how fast and far the electrical energy from a battery can move a boat.
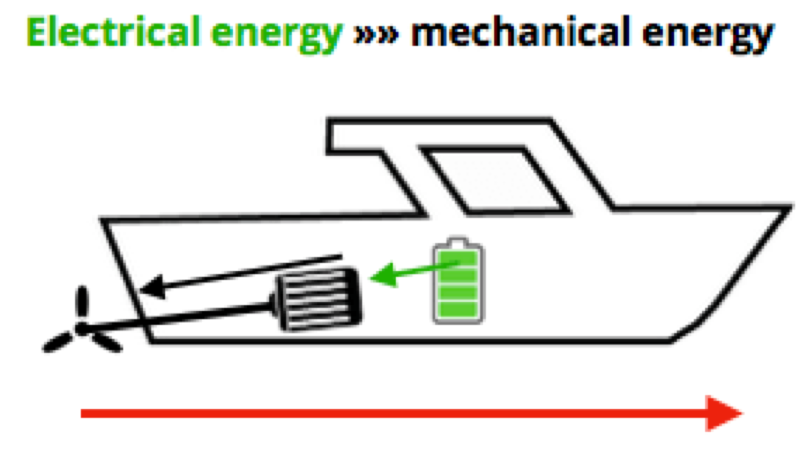
In terms of your boat, the energy is stored in your battery, that electrical energy is transferred to mechanical energy — the motor and drive train and propeller — which put force against the water to do the work of moving your boat. As we said, higher speed and/or bigger weight require more power transferred from electrical energy to mechanical energy — all measured in the same Watts.
We know the formula for mechanical power is Power = Work ÷ Time. There is also a specific formula for calculating electrical power: Watts = Volts x Amps.
To be clear, a watt is a watt is a watt, it doesn’t matter if it is measuring the rate of work of electrical power or mechanical power or if it is being exerted by an electric motor or fossil fuel motor, steam motor, or hand-cranked.
With electric boats, since the power of the batteries and the power of motors are both measured in Watts, it is fairly simple to match a battery with a motor and also figure out how much work it will be able to do for what amount of time.
A kiloWatt is 1,000 Watts
A Watt is the amount of work one Joule of energy can perform in one second, which is not much. Individual Joules and Watts are small. To give you an idea of how small a Joule is, a common unit of energy we all use is the food calorie, and a joule is about 1/4000th of a calorie – 4,184 Joules in a calorie. You burn about 2,000 calories a day, so you burn about 8,368,000 Joules a day (congratulations)
Because a Watt’s energy is so small, we often use kiloWatts to refer to any sizable amount of power. A kiloWatt (denoted as kW) is the work that one thousand Joules can do in one second.

How Much Horsepower?
Many people are used to the term horsepower when talking about boat engines. And, during this transitional time between ICE (internal combustion engines) power and electrical power, boaters will naturally want to know the equivalent horsepower of the electric motor. The answer is 1 kiloWatt is equal to 1.34 horsepower.
So, for an electric motor to produce 100-hp it must be continuously rated at 134 kiloWatts. The converse is a 100-kW motor has about 74.6-hp.
However, if you are going to be looking at or using an electric boat, I think you would be well advised to forget about horsepower and think only in terms of Watts, or even better, kiloWatts. In case you haven’t noticed, more and more engine companies in the last 10 years or so, have been giving their power ratings in both horsepower and kiloWatts. The term “horsepower” will soon go the way of, well,…the horse and buggy.
Battery Power for Your Electric-Boat Motor

Working with Watts = Volts x Amps
Let’s put this common measurement to use as applied to electrically powered boats. You are transferring electric power to mechanical power to move your boat and they are measured in the same units. To get all of the benefits of a 10kW electric motor you need to have a battery with a voltage and amperage that can supply at least 10 kW — 10,000 watts.
In theory, it could be any combination: a 50V battery sending a current of 200A would do it, as would a 100V battery sending 100A. In real life, though, there are constraints, which you can see by taking it to the ridiculous — a 2V volt battery sending 5,000A would be like trying to run a boat off a cell phone battery.

To give you some real-life examples, an electric trolling motor of 1kW might be perfect for fishing from a small boat, so a 12V battery and a current of 50A could achieve that.
At the other end, there are now electric outboard motors rated at more than 150kW and inboards well beyond that. To get high power, you need high voltage, high current, or medium both. In terms of amps, you’re getting up there when you’re talking about 300 amps and electric-car motors generally have voltages of 360V to 400V and up to 800V. If you had an 800V/300A motor it could put out up to 240,000 watts of power — 240 kW, while a 600V/200A motor could produce 120kW.
In the middle range of electric boat motors, the voltage is usually somewhere between 48V and 144V and the amps could go up to 250A or 300A.
When we’re talking about this equation of Watts = Volts x Amps and theory versus reality, there are some other things to be aware of. First is that motors have an input power and an output power. If you put in 10kW of power, some of it is used up in spinning the motor and some energy is lost as heat, so the output power will be smaller than 10kW. Not a lot smaller, electric motors are very efficient. But smaller.
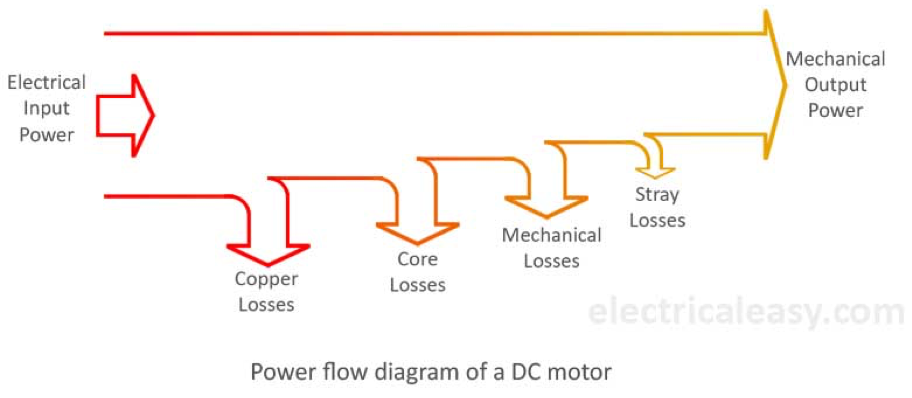
Motors may also have two power ratings. Peak power measures the maximum amount of power the motor has available, but only for short periods of time. Continuous power measures the power that can be put out at a constant rate for long periods of time. Some motor manufacturers only tell you one of the ratings, many tell you both.
Similarly, your battery may have a couple of ratings for the volts like the nominal voltage, voltage range, discharge voltage and others.
Power and Range — kiloWatts, kiloWatt hours, Amp hours

One of the nicest things about working in kiloWatts as the common measurement of both electric and mechanical energy is that it makes it easy to figure out much energy is required to keep your boat moving and how far it can go at what speed without recharging.
Calculating Your Range
With an understanding of kiloWatt hours and Amp hours, you can easily calculate that range.
kiloWatts, kiloWatt hours, Amp hours
The thing you want to know about your battery to make the range calculation is how many kiloWatt hours it has.
Note Carefully: kiloWatt hours, Not kiloWatts. KiloWatt hours are denoted as kWh.
The difference between kiloWatts and kiloWatt hours is important. A kiloWatt is a measurement of power, as we know. But a kiloWatt HOUR is a measure of energy.
How can that be? Let’s return to that other measurement of energy, the joule, for a minute.

We learned that a kiloWatt is the work that one thousand joules can do in one second and it follows that a kiloWatt Hour is how much work 1,000 joules per second can do in 1 hour – 3,600 seconds.
It is a measurement of energy because it tells us the number of units of energy being transferred in one hour to do the work: 1,000 joules per second x 3,600 seconds in an hour = 3,600,000 joules of energy converted to work in an hour.
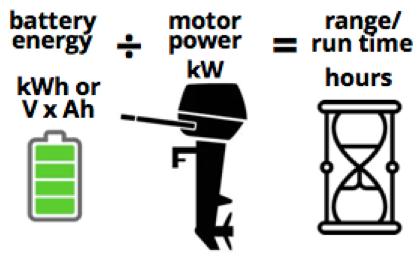
This makes it easy to know how long the energy in your battery can supply an accompanying motor. Simply divide the battery’s kiloWatt hour capacity by the motor’s kiloWatt power rating. A 10 kiloWatt motor – running flat out the whole time – needs 10 kiloWatts of electrical power each second and will use 10 kiloWatt hours of energy in 1 hour.
In other words, it will draw 3.6M joules of electrical energy out of the battery in an hour, when running at ‘wide open throttle’, to use a fossil fuel term.
Like any boat motor, though, a 10kW motor does not need to go at full speed and use all the peak power all the time. At half speed, it needs 5kW, at one-quarter speed it needs 2.5kW, etc.
That’s the good news about the calculation. The bad news is that not every battery manufacturer provides a specification for kiloWatt hours. Some do, some don’t.
However, almost all battery manufacturers provide a specification for Amp hours, designated as Ah.

You can still easily figure out the energy storage using amp hours, though. Just adapt the Volts x Amps = Watts equation to say Volts X Amp Hours = Watt Hours. (Be careful when calculating — the answer is in watt hours, not kiloWatt hours.)
Since amps are a measurement of how much electricity is flowing past a single point in the wire at any one time, an amp hour is how much flows past in an hour, how much energy is coming out of the battery.

Using our same 10 kiloWatt motor, it is going to use 10,000 watts in an hour (flat out), so with a 48V battery, we will need a specification of about 200 amp hours: 48V x 200Ah = 9,600 watt hours, which is just less than 10 kiloWatt hours. We can go at top speed for 1 hour, half speed for 2 hours, and quarter speed for 4 hours.
Bottom line is that estimating range with electrical power is just as easy as estimating range with internal combustion engines: Know how much fuel — or charge — you have, your energy use at a given speed (as with your mpg or gph with internal combustion engines) and the rest comes easy.
Battery Specifications

When you look at your battery specifications to find the amp hours and other specifications you may find a few references that could be confusing — different kinds of voltage, maximums and minimums for amperages, and other unfamiliar terms.
Voltage
Nominal voltage, for all intents and purposes, is the voltage of your battery. Nominal means ‘named’ — so it is the named voltage of your battery even though the actual measured voltage might be higher or lower. A 48-volt battery might actually be a 51.2V battery when analyzed, but it is easier for everyone to name it (and similar batteries) a 48V battery.

Most nominal 12V batteries like the one in your car or truck put actually max out at 13.8 volts, but they are still “12-Volt” batteries.
Voltage Range
Going back to the electricity as water analogy for a minute, when you draw electricity out of a battery, the ‘pressure’ fluctuates a bit — when you want to increase the power of your motor for instance – and overall it gradually goes down as more electricity is used. At a certain point, there is not enough ‘pressure’ to send the current to the motor.
So the voltage range will give you the maximum voltage you can expect and the minimum voltage. For example, a battery with 48V nominal might have a voltage range of 58.4V to 40V.

Minimum Discharge Voltage
This is the minimum voltage when there is not enough voltage to get the current flowing.
Charge Voltage
This is the voltage the charger will use. If you are buying a battery with a compatible charger you won’t need to worry about it. Almost all electric boat batteries can be charged using a household current (Level 1 charging) and many are designed to be compatible with standard Level 2 electric vehicle fast chargers.
Amperage
Continuous amps will tell you the electric current that can flow out of the battery on a continual basis, the ‘cruise’ amperage.
Maximum amps will tell you the maximum electric current that can flow out of the battery. When you need more power to get through waves or go faster, you will need more amps: Watts = Volts x Amps.
Charge amps or charge current is similar to the charge voltage. It is the current that will flow back into the battery to recharge it, but you will not need to worry about it with a compatible charger.

Amp hours we have already covered, it is the energy storage capacity of the battery.
kiloWatts
Some batteries may specify the kiloWatts, the power rating. Similar to peak power or maximum amps, it is the power the battery can deliver at any single point in time.
kiloWatt hours may be specified as the storage capacity on some batteries but as noted earlier, Ah is the more common designation and can be easily converted to kWh through Volts x Ah = kWh.
Be careful on this — sometimes even manufacturers (or the people in their labeling department) will confuse kW with kWh.
You will also find other specs about your battery like operating temperature range, number of cycles and other things, but these are pretty self-explanatory.
How Batteries Work

This seems as good a time as any to look into how batteries work. They all basically work the same, though the internal chemistry for storage may be different. A note to all experts out there, this is extremely simplified.
A battery has one kind of material with atoms that want to shed their electrons, another type of material with atoms that want to gather electrons, and in between them a material that facilitates a chemical reaction that sets the electrons free so they can go and collect on the other material.
The Simplest Battery
As a kid you may have made a battery made out of a lemon, a nail and a copper penny. The zinc in the galvanized nail wants to get rid of electrons, the copper in the penny wants to gather them, and the lemon juice is the material in between.
The nail and the penny are called the electrodes of the battery. The nail is the negative electrode — the anode — the penny is the positive electrode — the cathode — and the lemon juice is called the electrolyte.
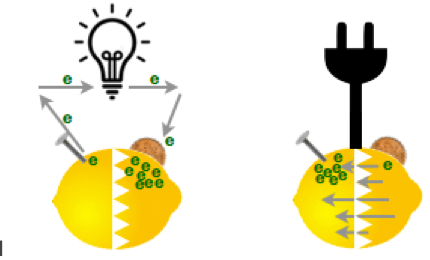
In a rechargeable battery, like the kind you want in your boat, the chemicals are a lot more complicated and there is also a one-way barrier. Hooking up a motor between electrodes creates a chemical reaction with the electrolyte that makes the electrons flow through the motor to get from the anode to the cathode (this is called electricity) and the motor spins. When you plug it into a recharger, the one-way barrier lets the electrons go directly back through the barrier to the other electrode.
As you can imagine, the chemical reaction of a lemon, nail and copper coin doesn’t generate a lot of electricity. Surprisingly, neither do the chemical reactions of the latest and most efficient battery chemistries — it’s only about 2 volts to 3.6. volts.
We’ll learn more about how they produce lots more voltage than that in the next sections.
Battery Cells, Modules and Packs

The way to get greater voltage is to connect more batteries. But the word ‘battery’ is imprecise. We use it to mean everything from something in a hearing aid to something in an electric vehicle.
Proper Terminology
To be more precise, the lemon is a battery cell. Battery cells are connected together to make modules and modules are connected together to make battery packs. Even some of the batteries you use at home, like a 9V battery, are technically battery modules constructed of six individual 1.5V battery cells.

Here’s a crazy example of how this works — the world’s largest lemon battery — that demonstrates how a battery pack is created to increase voltage.

On a more practical level, the battery for a Tesla 3 (or any other electric vehicle) is not much different. It uses lithium-ion cells, each a little bigger in dimension than AA batteries. Each cell can generate 3.7 Volts and there are 2,976 of them arranged in 96 groups of 31 to create the Tesla 3 battery pack of 350 volts. Energy storage capacity of the pack is 80 kWh. You can see the cells, modules and overall pack in the photo below.

Strength in Numbers
The Tesla battery (and all batteries) use two different ways of connecting batteries to achieve both higher voltage and higher kiloWatt hours.
If battery cells or modules are connected in series, the voltage is increased, but the energy storage is not increased.
If they are connected in parallel, the opposite happens — energy storage capacity is increased but the voltage stays the same.
In the case of a large battery like those used in electric cars and larger electric boat systems, the work is already done for you. For smaller motors, though, you can do this yourself. Two 48V batteries (for instance) can be put together in series to drive a larger motor. Or, they can be put together in parallel to double the range of your boat.
IMPORTANT NOTE: This is not true for all electric motors and batteries — do NOT do this without consulting the manufacturers.
Different Cells: Cylinders, Pouches, Prismatic

One final thing about how batteries are built. For lithium batteries there are three basic types of cells — cylindrical, pouch, and prismatic.
Cylindrical cells are similar to AA batteries. Prismatic cells are contained in a rectangular can. Pouch cells are small pouches.
Each cell type is constructed in a different way and has advantages and disadvantages for the manufacturer and user. These are mainly related to efficiency, weight and cost — in the manufacturing of the cell itself and in connecting them to make modules and packs.
Battery Management Systems

We know that a boat has different power needs and draws the appropriate current from the battery pack, but does the current come from one cell at a time or from all the cells at once or somewhere in between? That’s where a Battery Management System (BMS) comes in.
Peace of Mind
The BMS can rightfully be considered the brains of the operation. It monitors and manages the battery during discharging and charging, balancing the voltage and current of the cells so that the work of the pack is shared equally and safely.
Overall, its function is to protect the battery — and you — by constantly monitoring every cell in the pack and calculating how much current can safely go in and how much can safely come out. If you think of the minimum and maximum voltages and amps in the battery specifications, they apply to the battery pack as a whole and to each individual cell.
Through this monitoring, it acts as the battery’s fuel gauge and calculates the overall state of charge — how much energy is remaining in the battery pack.
It also monitors temperature and checks for indications of any loose connections, short circuits or insulation problems in the cells, modules and pack.
If the BMS detects anything unsafe in the operation, it will shut down the battery. It will also shut down the battery if it detects activity like excess or deficient voltage that could damage any cells. Aside from safety, this also extends the lifespan of the pack.
The BMS is usually a separate piece of equipment, but can also be built into the pack itself, in which case the pack is referred to as a smart battery.
Battery Chemistry

When discussing battery chemistry, we are referring to the materials involved in the chemical reaction that produces the electric charge. There are two base materials used for the rechargeable batteries you will want for an electric boat: Lead and Lithium. Within each type of battery, there are many options.
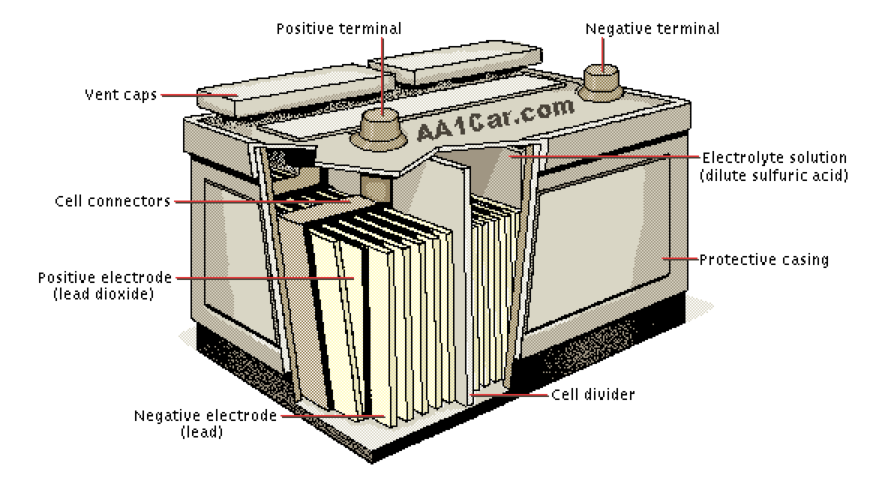
The chemical reaction in lead batteries takes place between electrodes made of lead (the anode is lead metal and the cathode is lead oxide) and an acid. Hence the name lead-acid battery.
Lithium batteries use a variety of materials and alloys to cause the chemical reaction. The specific combination is usually available in the user manuals and brochures, but is often not advertised. The generic Lithium-ion or Li-ion is used.
The one type of lithium battery that usually has the specific chemistry in the name is the LiFePO or LiFePO4 battery, which is not lithium ion but lithium iron phosphate.

For lithium batteries there is a six-point system that gives ratings on:
- Specific Energy
- Specific Power
- Safety
- Performance
- Life Span
- Cost
We have used the system to provide a basis of comparison for the most common lithium chemistries and have adapted it slightly for lead-acid batteries to give an indication of where they fit in the scheme of things.
The 6 Rating Criteria
Specific Energy can also be called gravimetric energy density and refers to the amount of energy that can be stored in the battery by weight: watt hours per kilogram.
The term energy density is often used, but isn’t entirely accurate because energy density actually refers to how much energy can be stored by volume: kWh per liter. This by-volume spec is rarely used in consumer information, so if you see energy density it probably (but not always) means the by weight specific energy specification.

A high specific energy or energy density is better: It translates to a lighter battery. For lithium batteries this number could be anywhere from 100 watt hours per kilogram to 250 watt hours per kilogram, depending on the chemistry. Specific energy ratings for lithium batteries are 100 to 250 watt hours, not kiloWatt hours.
The lithium rating in kWh would be .1 to .25 kWh per kilogram of battery material. Lead acid batteries have specific energy of about 35-40 watt hours per kilogram (.03 kWh/kg).
The numbers for lithium refer to the energy of an individual cell. The energy density of the battery pack as a whole will be lower because it needs to take into account the weight of all the connections and coolants and glues that go into constructing the pack. The lead acid numbers are for the battery pack.

Specific Power is the amount of power per weight that the chemistry can deliver: kW per kilogram.
This is easy to understand now that we know the difference between energy and power. Lithium batteries can range from about 250 watts per kilogram to 350 w/kg. Lead acid is about 180w/kg.
Safety. This specification on the chart is the safety compared to other lithium batteries.
When it comes to concerns about battery safety, some people point to news reports of electric-vehicle fires. The reason there are news reports about EV battery fires is the same reason there are news reports about airplane accidents. They are rare. Nobody reports on the millions of hours of battery use that go on every day with no incident.

There is certainly some risk involved in using lithium-ion batteries, just as there is some risk involved in using the volatile, explosive liquids that run fossil fuel motors. When batteries are used and maintained properly the risks are low. They are mitigated by thousands of hours of research and testing as well as the battery management system monitoring that goes on every second the battery is in use.
Performance in the graphs relates to performance at hot and cold temperatures.
Life span is an indication of the overall life span of the battery and specifically how many charging cycles it can go through.
Every discharge and recharge — called a cycle — results in the battery not being able to get all the way back to fully charged. Eventually, the battery is not useful. More cycles is better because it means your battery will last longer.
Determining how many cycles a battery can undergo is difficult because the definition of a cycle isn’t absolute in terms of how much energy is discharged and recharged to make up ‘a cycle.’
In general, though, lithium-ion batteries in electric vehicles and boats are designed to undergo 1,000 to 2,000 charge cycles.

Cost refers to relative initial cost compared to other lithium batteries. A note on looking at the graphs: the ratings show the desirability of the battery characteristics, so a low cost is higher on the rating scale because it is a good thing in a battery.
Lead Acid
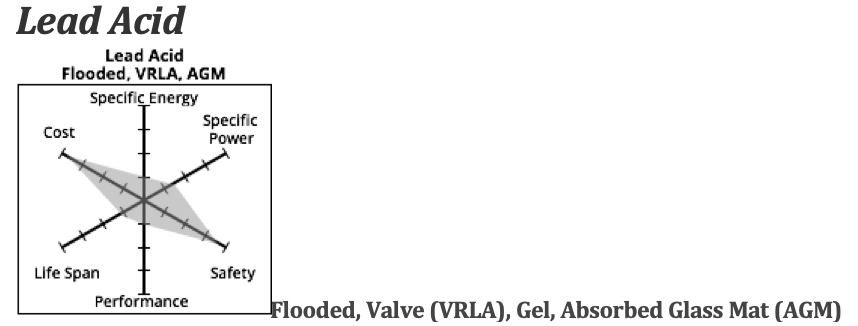
A starter battery is only used to start a motor, which requires more power, but for a short period of time: small voltage and high maximum amps. It doesn’t need big energy storage. It uses a small discharge to turn the massive weight of the fossil fuel motor, then the alternator (which is just a small electricity generating turbine) recharges the battery through the spinning of the fossil fuel motor.
For an electric boat, we want a battery that has a power output (Volts times Amps) compatible with the motor and lots of available kiloWatt hours to fully discharge the battery (up to about 80%) pulling electricity out of it for a long time.
This is called a deep cycle or sometimes a deep-discharge lead-acid battery.
There are a few types of lead acid deep cycle batteries — flooded batteries and valve regulated lead acid (VRLA). The VRLAs are further divided into gel batteries and Absorbed Glass Mat (AGM) types.
In the flooded versions the electrolyte is liquid, in gel versions it is a gel and in AGM it is held in a semi solid state by being held in the pores of the glass mat.
Flooded are the heaviest and least expensive…AGM the lightest and most expensive, but still much cheaper than lithium batteries.

Compared to lithium batteries, Lead Acid batteries have lower specific energy, lower cycles and lower initial cost. The specific energy is about half of the highest-density lithium batteries — and the specific power is about half that of lithium batteries. They generally are safe, however the flooded versions can release hydrogen gas and explode under certain conditions, especially if the owner has been a bit lax in maintenance.
The initial cost for Lead Acid is about 1/4 the price of a lithium battery, but since they last for only 300-400 cycles, the cost per cycle is higher. They are good for smaller motors, but after about 48 volts, the amount of power needed to move the weight of the battery pack alone makes it an impractical choice.
Lithium
There are many lithium-battery chemistries, but some are used for things like laptops and medical devices.

Below are the main varieties used for electric boat propulsion. The chemical composition refers to the materials used in the cathodes.

NMC is the most popular chemistry for electric powertrains in EVs, e-bikes and electric boats. The main reason for its popularity is that it has a high specific energy. Different manufacturers have their own recipes for the nickel, manganese and cobalt proportions so some will sacrifice a little specific energy for more specific power and vice versa. Battery manufacturers are trying to use less cobalt because of cost and concerns about ethical mining. More nickel means higher energy density, lower cost and longer cycle life, but a slightly lower voltage.
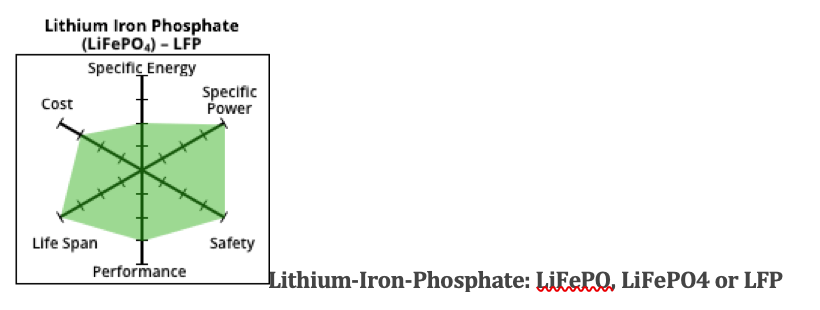
Many electric-boat owners and companies prefer the LiFePO lithium iron battery chemistry to lithium ion, largely because of lower cost and concerns about the safety of li-ion.

They make a tradeoff in range, though, because LiFePO has a low specific energy. It may not be the best battery choice for a high-speed, high-power electric boat. It is, however, becoming increasingly popular with sailboat owners where the increased battery weight in relation to the size of the boat is less important and speed under power is a non-issue.
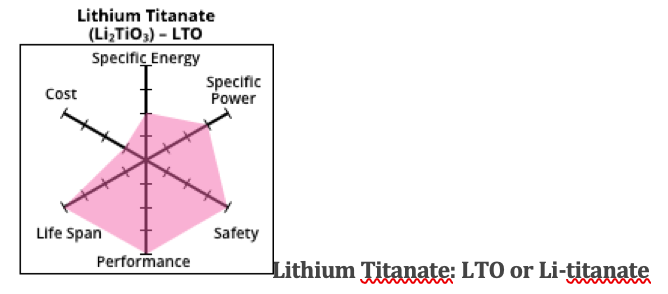
Lithium Titanate is a good chemistry for certain large vessels, but is not a popular option for recreational boats. It has a low specific energy — so batteries are heavy. On the other hand, they have long life span, are safe and can be charged quickly. The other drawback is their expense.

For a big electric ferry where the weight of the battery is less important but quick charging and public safety are important, the higher initial cost is offset by the long life span. This means the operator is free from the ongoing high operating costs of constantly filling up with fossil fuel.
Charging Your Lithium Battery

In the final installment of our 10-part tutorial on electrically powered boats, we will discuss the basics of charging and why the charger should be made specifically for lithium battery applications.
Conventional Battery Chargers
Almost all batteries for electric boats can be charged through a standard household circuit (Level 1) and almost as many have fast-charging capabilities (Level 2). In both cases the chargers use the AC electricity coming from the general grid and the technical aspects fall in line with charging for electric vehicles.
Standard existing charging pedestals at marinas can be used to charge electric boat batteries and there are DC high-speed charging units (Level 3) being installed in marinas around the world.

The pace of installation of these high-speed units should increase in the coming years as more people start using electric boats and higher-power electric-boat motors become more popular.
The pace of installation of these high-speed units should increase in the coming years as more people start using electric boats and higher-power electric-boat motors become more popular. Needless to say, this will take a major infrastructure investment by marina owners, something that the huge conglomerate marina companies will be able to manage, but will not be so easy for ma and pa operators.
What Chargers Do. The role of the battery charger is to take power from a source, usually 115V or 220V, and convert it to 12, 24 or 48V DC, depending on the voltage of the batteries, and charge them. The rule of thumb in the marine industry is to select a battery charger that has a capacity of about 10% of the batteries being charged. So, if it much charge a 200Ah battery a 20 amp charger is often fitted.
It is possible to charge a lithium-ion battery with a conventional charger, but it is not recommended and is fraught with problems. For example, a lead-acid battery will be fully charged at from 16.6V to 12.8V. A resting lithium-ion battery will not be fully charged until 13.3V-13.V. Because lead-acid chargers must use a lower voltage to charge batteries, they can only fill around 80% of a lithium battery. In the process, a lithium battery will be denigrated, among other problems.
Lithium-Ion Battery Chargers
The voltage from the source (115V-220V) cannot be higher than what the charger can handle, and the output voltage must be compatible with the battery. Voltage regulation by the charger is vital because lithium batteries are sensitive to over-voltage. The best units employ thermal protection to keep chargers at a safe temperature. Further, it must provide Constant Current/Constant Voltage (CC/CV) management as required by the battery maker.

5x Faster. I lithium battery charger will chare the proper matched batteries five time faster than a lead-acid battery charger. Also, a lithium battery charger will charge the battery no matter what the battery’s charge is.
Most of the electric propulsion boats, and outboard motors, we have seen provide a complete system of charger, battery, inverter and motor and married together. That way the system’s engineers have made sure all components are as efficient, compatible and safe as possible.
Conclusion
This series article is intended to give you a basic understanding of batteries for electric boat propulsion. There are other things that could have been added, but the intent was to try to stay with the basics.
To stay as concise as possible, it also deals with the underlying mathematics of the battery/motor relationship without too many caveats about real life. For example, if a person has a 10kW motor and 10kWh battery it may not run full out for exactly an hour. The battery management system (BMS) won’t let the battery discharge 100%, and even if it did the motor may not be exactly 10kW and there are lots of other variables. But it will run for about an hour — the only question is how fast it will propel the boat.

If you are looking for more in-depth battery knowledge, an excellent resource is the Battery University website. And if you would like to find out more about the electric motors available for your needs, check out the Plugboats Guides to Electric Outboards under 5kW, Electric Outboards over 5kW, Electric Inboards, Electric Saildrives and Pods and Electric Trolling Motors.